-
在使用电烙铁进行焊接工作时,焊接处会持续产生烟雾。由于焊接处有铅、锡、铜等金属,那么在焊接时这些金属可能会以化合物的形式存在于烟雾中,造成局域空气污染,对人体的健康造成危害[1]。因此,对焊接处的烟雾进行快速检测意义重大。
目前基于化学方法检测烟雾的方法是通过收集器收集烟雾,得到样品,用各种试剂检测烟雾成分,没有办法实现原位、多元素在线检测。同时,由于电烙铁焊接锡线产生的烟雾中重金属污染是微量且实时的,对化学方法检测的精确性和实时性提出了很大的挑战。现阶段,关于电烙铁焊接锡线时产生的烟雾检测方面的研究尚鲜见报道。而基于光学技术的激光诱导击穿光谱技术有望实现对烟雾的在线检测[2]。激光诱导击穿光谱技术(laser-induced breakdown spectroscopy,LIBS)是一种对等离子体发射光谱进行分析以确定样品的成分及含量的技术。该技术使用强激光作用在样品表面,使其电离产生等离子体。作用结束后,在等离子体冷却过程中,处于激发态的原子和离子向低能级跃迁,产生特征谱线。分析这些特征谱线的关系就能得到样品中元素种类和浓度信息。LIBS技术具有多元素同时分析、样品损失小、实时在线探测、无接触等优势,在检测固体[3-6]、液体[7-11]、气体[12]和气溶胶[13]方面有很大的潜力。
目前使用LIBS检测气体和气溶胶的研究工作很少,KHUMAENI等人[14]用横向激发大气激光诱导击穿光谱技术分析钠气溶胶。GUO等人[15]使用LIBS技术成功地探测了大气气溶胶中Cr元素。PHONGIKAROON等人[16]使用LIBS对气溶胶中的重金属铈和钆并进行定量分析。这些研究虽然实现了采用LIBS对气溶胶中的元素进行定性和定量的分析,但是没有充分发挥LIBS的实时性和原位性的技术优势。本文中在自行搭建的实验系统的基础上, 成功实现了对电烙铁焊接锡线的烟雾进行在线原位检测。
-
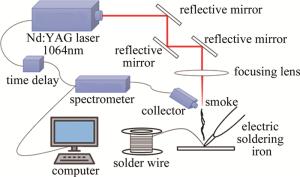
基于LIBS的实时检测实验系统如图 1所示。由Nd: YAG脉冲激光器、自行搭建的反射镜和对焦透镜组成的光学系统、光谱仪(配备充电耦合装置)、触发装置、时延装置、计算机、光纤探头组成。本实验中所用的激光单脉冲能量是260mJ,激光的基本波长是1064nm,频率为10Hz,持续时间为6ns,触发装置检测时间为1.5μs。
-
为了模拟工人使用电烙铁焊锡时的情况,实验时采用工人的手法,即低温上锡法。先将电烙铁插上电源,等待烙铁头加热至200℃后,用烙铁头沾松香(保护烙铁头),等待1min(待松香消失)后上锡,用锡线与烙铁头充分接触,使锡线融化在烙铁头上。在此过程中,烙铁头产生烟雾。不断调整烙铁头,使得产生的烟雾位于聚焦透镜焦点处斜下方。使用扇子朝烟雾附近略微扇风,引导烟雾飘向透镜焦点。激光透过透镜垂直聚焦在烟雾表面形成焦点,从而在烟雾表面激发出高温等离子体,使用光线探头收集粒子跃迁时产生的光谱,通过光纤导入光谱仪中记录数据,最后导入计算机进行分析。
-
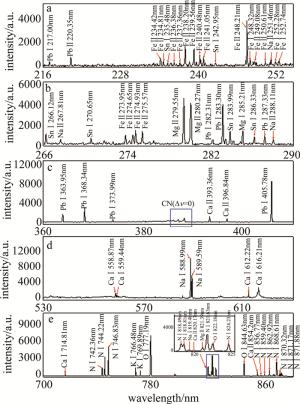
经过多次实验, 对比选取了具有代表性的LIBS光谱。为了解决光谱数据不同波段波长漂移现象,减少实验误差,在标定元素的特征谱线前,通过美国国家标准与技术研究院(National Institute of Standards and Technology, NIST)数据库中不同元素的特征谱线的波长与本实验中所得的光谱数据对比,校准光谱波长的漂移。
校准后,对216nm~255nm,266nm~290nm,360nm~410nm,530nm~630nm,700nm~880nm波段的光谱进行元素标定,标定结果如图 2所示。经过整理,得出了电烙铁焊锡烟雾的光谱谱线的鉴别表,见表 1。烟雾中观察到Sn, Pb, Fe, Na, K等金属元素,还检测到N, O, C等非金属元素。
表 1 Characteristic spectral lines of the main elements in smoke
element characteristic spectral lines/nm Sn 235.48, 242.95, 270.65, 283.99, 286.33 Pb 220.35, 283.30, 363.95, 368.34, 373.95, 405.78 Fe 238.20,239.56,248.32,274.93,275.57 N 742.36,744.22,746.83 Ca 393.36,396.98,558.87,612.22,616.21,854.2 K 766.48,769.89 Na 588.99,589.59 Mg 279.55,280.27,285.21 O 777.19, 844.63 在363.95nm, 368.34nm, 373.99nm, 405.78nm处发现Pb的特征谱线,并满足NIST数据库中实验得出的铅元素谱线强度关系,验证了电烙铁焊接含铅锡线时产生的烟雾中含有重金属铅的猜想。进行电烙铁焊接时需要精细操作,这意味着人们需要靠近焊接点,则铅元素会通过人们的呼吸道进入人体,不仅影响人体血红蛋白的合成,诱发溶血,而且破坏消化系统的粘膜,造成萎缩性胃炎[17-18]。
-
仅仅从使用含铅锡线的电烙铁工作产生的烟雾中检测出铅元素还不能说明含铅锡线对工作人员健康的危害,实验中要对烟雾中的铅元素进行定量分析,大致估算出烟雾中铅元素的含量,以此来证明使用电烙铁焊接含铅锡线的危险。实验中采用的是元素内标法[19-20]。
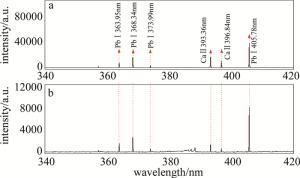
将样品分成3份浸泡在不同质量分数的(CH3COO)2Pb·3H2O溶液中,在40℃的低温下烘干。经过计算,3份样品中铅元素的质量分数为5×10-5,10×10-5,20×10-5。通过LIBS技术能够得到各个样品的光谱。以含铅元素质量分数为20×10-5的样品的光谱为例:通过比较添加质量分数为20×10-5铅元素的样品与未添加任何物质的原始样品的光谱,选取340nm~420nm这一段光谱分析。如图 3所示。
由图 3可知, 363.95nm, 368.34nm, 373.99nm, 405.78nm这4条为铅的特征谱线。对铅元素的定量分析就由此为基础。
由Lomakin-Scheibe公式:
$ I=a w^b $
(1) 式中,I为谱线的观察强度, a是实验常数, w是目标元素的质量分数, b是自吸收系数。若忽略自吸收,即b=1,则(1)式可以改写为:
$ I=a w $
(2) 由于在不同的实验中,a的值是不同的, 但是使用内标法可以消除实验常数a对实验的影响。由于电烙铁烟雾中没有钙元素,且在不同实验中,钙元素是不变的, 因此,本实验中选取CaⅡ393.36nm为参考谱线,则公式可以改写为:
$ \frac{I_{\mathrm{Pb}}}{I_{\mathrm{Ca}}}=\frac{a_{\mathrm{Pb}} w_{\mathrm{Pb}}}{a_{\mathrm{Ca}} w_{\mathrm{Ca}}} $
(3) 进一步可化简为:
$ I_{\sum i}=A w_{\mathrm{Pb}} $
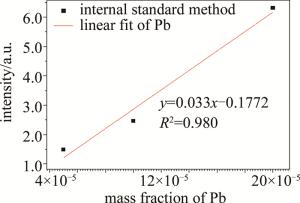
(4) 式中, I∑i为4条铅元素特征谱线的相对光强的和,wPb为铅元素的质量分数,A为常数。以I∑i为y轴,以wPb为x轴,用不同质量分数的样品重复实验,得到图 4。由图 4可知,拟合曲线与特征谱线光强有较好的线性关系,因此根据定标曲线,结合实时的空气颗粒物质量分数, 即可得到空气中的铅元素质量分数。
-
检出限(limit of detection, LOD)是样品中检测出待测物质的最小质量分数,是评价一个检测方法的重要指标。本实验中使用的元素内标法对待测样品中铅元素的含量有一定的要求,若铅元素质量分数过小,会导致背景光与样品中铅元素的特征谱线混淆,增大实验误差。因此,通过LIBS的检出限公式,结合铅元素的定标曲线,计算出铅元素的检出限:
$ L_{\mathrm{LOD}}=3 \sigma / K $
(5) 式中,σ是340nm~350nm波段多次实验求得的背景光强度的标准差, K是上文计算的铅元素定标曲线的斜率。经过计算,得出烟雾中铅元素的检出限为19.35×10-5。
-
当等离子区域需要满足局域热平衡(local thermal equilibrium,LTE)态,即粒子需要满足Boltzmann分布时,样品中的铅元素质量分数与其特征谱线的强度才回近似线性关系。因此,为了验证本实验对铅元素进行定量分析的可行性,需要判断等离子区域是否在实验中处于LTE态[21]。
利用McWhirter准则:
$ n_{\mathrm{e}} \geqslant 1.6 \times 10^{12} \times T^{\frac{1}{2}} \times \Delta E^3 $
(6) 式中,ne代表等离子体的电子数密度(单位为cm-3),T代表等离子温度(单位为K),ΔE代表所选相关元素相邻能级间最大能级差(单位为eV)。当实验光谱满足(6)式时,光谱有效。
-
使用Boltzmanna直线法可以计算出(6)式中的T。由Saha-Boltzmann方程[22]:
$ \ln \left(\frac{I_{i j} \lambda}{A_{i j} g_k}\right)=-\frac{1}{K_{\mathrm{B}} T} E+\ln \left[\frac{h c N_s}{U_s(T)}\right] $
(7) 式中, λ和Iij分别表示特征谱线波长和强度,Aij是原子或者离子的跃迁几率,gk是电子或者离子跃迁至上能级的统计权重,k表示上能级的序号, KB为Boltzmann常数,T是等离子温度,E是电子或者是离子跃迁至上能级所需的激发能,c是真空中的光速,h是Planck常数,Ns是电子数密度,Us(T)是电子或者是离子的配分函数, 下标s是电离次数。
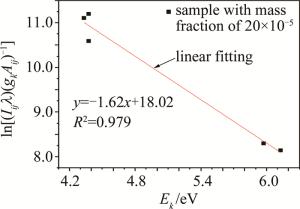
选取铅元素质量分数为20×10-5的样品,根据LIBS中的357.27nm,363.95nm,368.34nm,373.99nm,405.78nm这5条为铅的特征谱线的波长λ和强度Iij,在NIST数据库中查寻这5条铅元素特征谱线各自跃迁的机率Aij、跃迁至上能级的统计权重gk和激发能E,通过线性拟合的方法得到等离子温度T。如图 5所示,经过计算得温度T=7143K。横坐标Ek表示上能级能量。
-
Stark展宽是电子能级在外电场作用下发生能级分裂而出现的能级展宽,与电子数密度有关。因此可以通过谱线的半峰全宽w0和电子数密度ne之间的关系求得ne:
$ w_0 \approx 2 \omega \frac{n_{\mathrm{e}}}{10^{16}} $
(8) 式中,ω表示碰撞展宽系数。选取405.78nm铅元素的特征谱线,得到半峰全宽为0.295nm,计算得电子数密度为1.68×1017cm-3。
实验中,5条铅元素特征谱线中最大能级差ΔE=3.4693eV,等离子体温度T=7143K,得出电子数密度阈值为8.038×1015cm-3,小于1.68×1017cm-3。因此,实验中的等离子体满足(6)式,即本实验中得到的光谱为有效光谱。
-
使用基于LIBS技术的自行设计的实验系统对电烙铁焊锡线时产生的烟雾进行在线分析,在烟雾中发现多种金属元素(包括重金属元素铅),说明了使用含铅锡线会导致局域重金属空气污染。本实验中以CaⅡ393.36nm为参考谱线,使用元素内标法对铅元素进行定量分析得到了定标曲线,该曲线的线性相关系数为0.980,并计算出铅元素的检出限为19.35×10-5。为了验证光谱的有效性,使用McWhirter准则对实验光谱进行判定。对20×10-5的样品光谱中的5条铅元素的特征谱线进行线性拟合,通过Saha-Boltzmann方程计算出实验中等离子温度为7143K。通过Stark谱线展宽法估算出电子数密度1.68×1017cm-3,把计算出的等离子温度和电子数密度代入不等式,满足McWhirter准则,验证了实验光谱的有效性。本次实验说明, 使用电烙铁进行焊锡操作时,若使用的锡线含铅,则产生的烟雾中也会有重金属铅的存在,对室内空气造成污染,对使用者的健康造成危害。此外,本次实验证明了自行设计的LIBS实验系统在检测局域空气污染方面具有实时性和原位性的优势。
利用LIBS技术对电烙铁的烟雾进行在线分析
Online detection of smoke from the electric iron by LIBS
-
摘要: 为了研究电烙铁工作时焊接含铅锡线产生的烟雾对局域空气的污染, 设计了一套基于激光诱导击穿光谱技术(LIBS)的实验系统, 对电烙铁焊接含铅锡线的烟雾进行在线分析, 在烟雾的光谱中发现了重金属元素铅的特征谱线。使用元素内标法对铅元素进行定量分析, 通过分析铅元素的等离子体温度与电子数密度的相关特性, 验证了实验所得光谱的有效性。结果表明, 通过拟合曲线获得铅元素的检出限为19.35×10-5; 对比传统化学的化验方法, 基于激光诱导击穿光谱的电烙铁焊接锡线的场景检测实验系统和方法具有在线、原位、快速的优越性。该研究对解决电烙铁工作时室内空气的污染, 减轻对使用者健康造成的危害是有帮助的。Abstract: In order to explain the local air pollution caused by the smoke produced by soldering leaded tin wire with electric soldering iron, an experimental system based on laser induced breakdown spectroscopy was designed to analyze the smoke produced by soldering leaded tin wire with electric soldering iron, and the characteristic spectral line of heavy metal lead was found in the smoke spectrum. Lead was quantitatively analyzed by internal standard method, and the detection limit of lead was 19.35×10-5 by fitting the curve. By analyzing the correlation between the plasma temperature and electron number density of lead, the validity of the experimental spectrum was verified. The results show that the experimental system and method of scene detection of electric soldering tin wire based on laser-induced breakdown spectrum have advantages of on-line, in-situ and fast when compared with the traditional chemical test method.
-
Key words:
- spectroscopy /
- laser-induced breakdown spectroscopy /
- quantitative analysis /
- smoke
-
表 1 Characteristic spectral lines of the main elements in smoke
element characteristic spectral lines/nm Sn 235.48, 242.95, 270.65, 283.99, 286.33 Pb 220.35, 283.30, 363.95, 368.34, 373.95, 405.78 Fe 238.20,239.56,248.32,274.93,275.57 N 742.36,744.22,746.83 Ca 393.36,396.98,558.87,612.22,616.21,854.2 K 766.48,769.89 Na 588.99,589.59 Mg 279.55,280.27,285.21 O 777.19, 844.63 -
[1] WEI Y H, HUANG Q Ch. The toxicological effect of lead on the human health and its measures of preventing[J]. Studies of Trance Elements and Health, 2008(4): 62-64(in Chinese). [2] ZHANG Y, ZHANG T L, LI H. Application of laser-induced breakdown spectroscopy (LIBS) in environmental monitoring[J]. Spectrochimica Acta, 2021, B181: 56-63. [3] PERSHIN S M, COLAO F, SPIZZICHINO V. Quantitative analysis of bronze samples by laser-induced breakdown spectroscopy (LIBS): A new approach, model, and experiment[J]. Laser Physics, 2006, 16(3): 455-467. doi: 10.1134/S1054660X06030066 [4] CHEN L, YOU L B, LUO X F, et al. Detection of Cd in table salt by LIBS technology[J]. Laser Technology, 2019, 43(1): 6-10(in Chinese). [5] GANG J, CHEN Y Q, YANG Y X, et al. Time-resolved high sensitivity signal detection of chrominum in aluminum alloy by laster-induced breakdown spectroscopy[J]. High Power Laser and Particle Beams, 2017, 29(9): 156-160(in Chinese). [6] HAN S K, PARK S H, AHN S K. Quantitative analysis of uranium in electrorecovery salt of pyroprocessing using laser-induced breakdown spectroscopy[J]. Plasma Science and Technology, 2021, 23(5): 107-114. [7] XIU J Sh, DONG L L, LIN Sh, et al. Research progress of laser induced breakdown spectroscopy and other atomic spectroscopy in engine oildetection[J]. Laser Technology, 2018, 42(4): 505-510(in Chinese). [8] MONCAYO S, MANZOOR S, ROSALES J D, et al. Qualitative and quantitative analysis of milk for the detection ofadulteration by laser induced breakdown spectroscopy (LIBS)[J]. Food Chemistry, 2017, 232: 322-328. doi: 10.1016/j.foodchem.2017.04.017 [9] WANG Y, ZHAO N J, MA M J, et al. Chromium detection in water enriched with graphite based on laser-induced breakdown spectroscopy[J]. Laser Technology, 2013, 37(6): 808-811(in Chinese). [10] BAK M S, MCGANN B, CARTER C, et al. Determinants of laser-induced breakdown spectra in N2-O2 mixtures[J]. Journal of Phy-sics, 2016, D49: 125202. [11] QUE Zh B, LU W X, XIAO W, et al. LIBS analysis of heavy metal elements in suspended particulate matter in the Yangtze River[J]. Laser Journal, 2019, 40(5): 31-34 (in Chinese). [12] YANG W B, LI B C, HAN Y L, et al. Quantitative analysis of trace oxygen concentration in argon and nitrogen based on laser-induced breakdown spectroscopy[J]. Chinese Journal of Lasers, 2017, 44(10): 1011001(in Chinese). doi: 10.3788/CJL201744.1011001 [13] ZHANG L, WANG Zh, DING H B. Application of LIBS in diagnosis of aerosol[J]. Journal of Atmospheric and Environmental Optics, 2016, 11(5): 338-346 (in Chinese). [14] KHUMAENI A, KURIHARA K, LIE Z S, et al. Analysis of sodium aerosol using transversely excited atmospheric CO2 laser-induced gas plasma spectroscopy[J]. Current Applied Physics, 2014, 14(3): 47-54. [15] GUO W L, QIU R, WANG Ch F, et al. Detection of chromium in atmospheric aerosol by laser induced breakdown spectroscopy[J]. Chinese Journal of Quantum Electronics, 2020, 37(6): 745-751(in Chinese). [16] PHONGIKAROON W S. Elemental detection of cerium and gadolinium in aqueous aerosol using laser-induced breakdown spectroscopy[J]. Applied Spectroscopy, 2016, 70: 1700-1708. doi: 10.1177/0003702816648327 [17] HU X, ZHANG Y, DING Z H, et al. Bioaccessibility and health risk of arsenic and heavy metals (Cd, Co, Cr, Cu, Ni, Pb, Zn and Mn) in TSP and PM2.5 in Nanjing, China[J]. Atmospheric Environment, 2012, 57(1): 146-152. [18] HU Z J, SHI Y L, NIU H Y, et al. Synthetic musk fragrances and heavy metals in snow samples of Beijing urban area, China[J]. Atmospheric Research, 2012, 104: 302-305. [19] MA C H, XIAO L. Optimization method of quantitative analysis of mn in molten steel based on LIBS[J]. Journal of North China University of Science and Technology (Natural Science Edition), 2016, 38(3): 9-13(in Chinese). [20] LIU Y, LU J D, LI P, et al. Application of internal standard method in the determination of carbon content in pulverized coal by laser induced breakdown spectroscopy[J]. Proceedings of Chinese Society for Electrical Engineering, 2009, 29(5): 1-4(in Chinese). [21] HUDDLESTONE R H, LEONARD S L. Plasma diagnostic techniques[M]. New York, USA: Academic Press, 1965: 201-264. [22] ASGILL M E, GROH S, NIEMAX K, et al. The use of multi-element aerosol particles for determining temporal variations in temperature and electron density in laser-induced plasmas in support of quantitative laser-induced breakdown spectroscopy[J]. Spectrochi-mica Acta, 2015, B109: 21-27. -

 网站地图
网站地图

 下载:
下载: