-
皮瓣移植手术是外科中常见的手术,皮瓣移植手术失败的主要因素是血管吻合处发生栓塞,栓塞会阻碍皮瓣内的血液循环引起皮瓣坏死[1]。术后若能尽早发现血管栓塞,对提高移植手术的成功率非常重要。目前术后护理主要通过医师的主观经验判断移植皮瓣存活状况,如测量皮温、观察皮瓣水肿程度、伤口渗出物的形状及颜色等,缺乏客观标准。因此,为皮瓣移植手术提供一种连续、无创而且简便实用的术后监测手段在临床上具有重要意义。
术后皮瓣存活的关键在于能否自我供血[2-5],利用近红外光谱可以无创、连续监测人体血运状况[6-7],它依据人体血液不同成分对近红外光吸收能力的差异,通过检测组织对近红外光的吸收程度,解算出人体局部组织的氧合参量。本文中通过检测术后创面部位的氧合参量,判断皮瓣血液循环情况,实现监测皮瓣存活状态的目的。为此本文中设计了组织氧饱和度实时监测系统,在组内进行了稳定性验证实验和前臂阻断模拟皮瓣血管栓塞实验,检验了该系统用于术后皮瓣血运状况监测的有效性。
-
人体组织中密布着大量的微血管, 包括微动脉、微静脉和毛细血管,人体组织的血氧参量即为上述各种微血管中血液血氧参量的加权平均[8]。因此,通过监测创面周围的组织氧饱和度可以达到反映术后皮瓣血运状况的目的。
局部组织氧饱和度(regional tissue oxygen saturation, rSO2)的定义为局部组织中氧合血红蛋白浓度CHBO2与局部组织中氧合血红蛋白浓度、还原血红蛋白浓度CHB和的比,有:
$ {R_{{\rm{rS}}{{\rm{O}}_2}}} = \frac{{{C_{{\rm{HB}}{{\rm{O}}_2}}}}}{{{C_{{\rm{HB}}}} + {C_{{\rm{HB}}{{\rm{O}}_2}}}}} \times 100\% $
(1) 式中, RrSO2为局部组织氧饱和度的值。在近红外波段(700nm~900nm),组织中还原血红蛋白(HB)和氧合血红蛋白(HBO2)是主要的吸光物质[9],本文中使用两种特定波长的光交替照射组织,在光源同一平面一定距离处接收组织的漫散射光。当波长为λ1,λ2的光穿过组织时,由吸收定律得:
$ \left\{ {\begin{array}{*{20}{l}} {{\mu _{{\rm{a}}, {\lambda _1}}} = {\varepsilon _{{\rm{HB}}, {\lambda _1}}}{C_{{\rm{HB}}}} + {C_{{\rm{HB}}{{\rm{O}}_2}}}{{\cal E}_{{\rm{HB}}{{\rm{O}}_2}, {\lambda _1}}}}\\ {{\mu _{{\rm{a}}, {\lambda _2}}} = {\varepsilon _{{\rm{HB}}, {\lambda _2}}}{C_{{\rm{HB}}}} + {C_{{\rm{HB}}{{\rm{O}}_2}}}{\varepsilon _{{\rm{HB}}{{\rm{O}}_2}, {\lambda _2}}}} \end{array}} \right. $
(2) 式中,μa, λ1是波长为λ1时组织的吸收系数,μa, λ2是波长为λ2时组织的吸收系数; ε是摩尔消光系数,只与吸收物质和波长有关。
由(1)式、(2)式得出局部组织氧饱和度为:
$ {R_{{\rm{rS}}{{\rm{O}}_2}}} = \frac{{{\varepsilon _{{\rm{HB}}, {\lambda _1}}} - {\varepsilon _{{\rm{HB}}, {\lambda _2}}}\frac{{{\mu _{{\rm{a}}, {\lambda _1}}}}}{{{\mu _{{\rm{a}}, {\lambda _2}}}}}}}{{\frac{{{\mu _{a, {\lambda _1}}}}}{{{\mu _{a, {\lambda _2}}}}}\left( {{\varepsilon _{{\rm{HB}}{{\rm{O}}_2}, {\lambda _2}}} - {\varepsilon _{{\rm{HB}}, {\lambda _2}}}} \right) - \left( {{\varepsilon _{{\rm{HB}}{{\rm{O}}_2}, {\lambda _1}}} - {\varepsilon _{{\rm{HB}}, {\lambda _1}}}} \right)}} $
(3) 从(3)式可以看出,求解RrSO2的实质是解出两个光源下被测组织的吸收系数之比。
人体组织对近红外光的吸收远小于散射,尽管单个光子在组织中的传播路径是随机的,但大量光子在组织中依然遵循统计规律,光子在人体组织中的传输路径大致呈月牙状[10-12]。由组织中近红外光谱的漫散射方程[13-14]得:
$ {\mu _{\rm{a}}} = \frac{{{{\left( {\frac{{\partial {T_{{\rm{OD}}}}}}{{\partial d}}\ln 10 - \frac{2}{d}} \right)}^2}}}{{3{\mu _{\rm{s}}}}} $
(4) 式中, ΤOD为光密度(optical density, OD),定义ΤOD=lg(I0/I), I0为入射光强, I为出射光强;d为光源到探测器中心的距离;μs为组织对近红外光的散射系数,与波长无关[15]。
当使用两束不同波长的光照射组织时,组织对两路光的吸收系数比为:
$ \frac{{{\mu _{{\rm{a}}, {\lambda _1}}}}}{{{\mu _{{\rm{a}}, {\lambda _2}}}}} = {\left[ {\frac{{\frac{{\partial {T_{{\rm{OD}}, {\lambda _1}}}}}{{\partial d}}\ln 10 - \frac{2}{d}}}{{\frac{{\partial {T_{{\rm{OD}}, {\lambda _2}}}}}{{\partial d}}\ln 10 - \frac{2}{d}}}} \right]^2} $
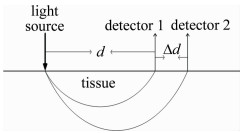
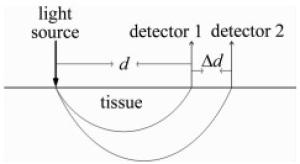
(5) 为求出$\partial {T_{{\rm{OD}}}}/\partial d$,在系统中引入第2个光电探测器,探测器于光源同侧一远一近分布,空间分布概念图见图 1。此时两波长的吸收系数比可简化为:
$ \frac{{{\mu _{{\rm{a}}, {\lambda _1}}}}}{{{\mu _{{\rm{a}}, {\lambda _2}}}}} = {\left[ {\frac{{\Delta {T_{{\rm{OD}}, {\lambda _1}}} - \left( {\frac{{2\Delta d}}{{d\ln 10}}} \right)}}{{\Delta {T_{{\rm{OD}}, {\lambda _2}}} - \left( {\frac{{2\Delta d}}{{d\ln 10}}} \right)}}} \right]^2} $
(6) 式中,ΔΤOD为同一波长下不同探测器探测到的光强差的绝对值;Δd为两探测器中心点的距离。结合(3)式,组织氧饱和度计算公式可化简为:
$ \begin{array}{l} {R_{{\rm{rS}}{{\rm{O}}_2}}} = \left\{ {{\varepsilon _{{\rm{HB}}, {\lambda _1}}} - {\varepsilon _{{\rm{HB}}, {\lambda _2}}}{{\left[ {\begin{array}{*{20}{c}} {\lg \frac{{{D_{1, {\lambda _1}}}}}{{{D_{2, {\lambda _1}}}}} - \left( {\frac{{2\Delta d}}{{d \cdot \ln 10}}} \right)}\\ {\lg \frac{{{D_{1, {\lambda _2}}}}}{{{D_{2, {\lambda _2}}}}} - \left( {\frac{{2\Delta d}}{{d \cdot \ln 10}}} \right)} \end{array}} \right]}^2}} \right\} \div \\ \;\;\;\;\;\;\;\;\;\;\;\;\;\left\{ {{{\left[ {\begin{array}{*{20}{c}} {\lg \frac{{{D_{1, {\lambda _1}}}}}{{{D_{2, {\lambda _1}}}}} - \left( {\frac{{2\Delta d}}{{d\ln 10}}} \right)}\\ {\frac{{{D_{1, {\lambda _2}}}}}{{{D_{2, {\lambda _2}}}}} - \left( {\frac{{2\Delta d}}{{d\ln 10}}} \right)} \end{array}} \right]}^2}} \right. \times \\ \left. {\left( {{\varepsilon _{{\rm{HB}}{{\rm{O}}_2}, {\lambda _2}}} - {\varepsilon _{{\rm{HB}}, {\lambda _2}}}} \right) - \left( {{\varepsilon _{{\rm{HB}}{{\rm{O}}_2}, {\lambda _1}}} - {\varepsilon _{{\rm{HB}}, {\lambda _1}}}} \right)} \right\} \end{array} $
(7) 式中,D1, λ1 ,D2, λ1 ,D1, λ2 ,D2, λ2 为远近探测器采集到λ1,λ2的光通过组织后的漫散射光强;摩尔消光系数ε通过查表可得。由此,通过光电探测器采集被测部位的漫散射光,便可进行被测部位RrSO2的估算。
-
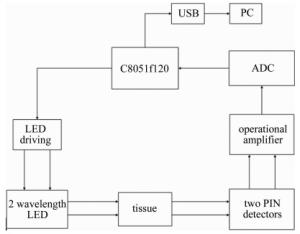
本文中提出的近红外光谱监测皮瓣移植术后状况系统如图 2所示。以C8051f120单片机为核心搭建测量系统。采用反射式传感结构,单片机驱动两路发光二极管(light emitting diode, LED)分时发出恒定强度的光,位于同侧的光电探测器采集通过人体组织的漫散射光,将光信号转变成电信号后通过运算放大器实现I-V(电流-电压)转换、放大。经模数转换器(analog-to-digital converter, ADC)转换后将放大后的模拟信号转变成携带血氧信息的数字信号,最后通过通用串行总线(universal serial bus, USB)接口实现上位机通信。
波长700nm~900nm的光对组织具有良好的穿透性,此波段下组织中的主要吸光物质为HB和HBO2,背景吸收干扰最小。如图 3所示,在750nm处和850nm处HB与HBO2的消光系数相差最大,因此选取750nm和850nm的光作为系统光源。为减小误差,使用集成双波长(750nm,850nm)发光二极管作为光源,共阳极设计。以ULN2003APG芯片为核心建立LED驱动电路,LED阴极连接可调变阻器调节入射光强。光电探测器放置在距离光源同侧10mm和20mm处[16]。
选用ADI公司的AD626芯片作为运算放大模块,将光电探测器接收到的光电流信号转变成与漫散射光强呈正比的电压信号并进行放大,使电路稳定的同时具有较高的信噪比。为保证两路探测器的信号模数转换同时进行、提高测量精度和转换速度,选用ADI公司的AD7060芯片搭载外围电路作为本系统的ADC转换模块,该芯片拥有16位ADC转换,8通道同步采样模数数据,具有精度高、同步性好、转换速度快的优点,满足实验需求。
目前国内外I-V转换大都通过运算放大器的外接电阻进行,本文中提出一种I-V转换思路:由于光电探测器可以理想化成一个恒流源与定值电阻的并联,为简化电路,在芯片外围电路中不跨阻,利用光电探测器内阻完成I-V转换。为验证两种方案测得的数据的准确性,分别将两种运算放大模块接入系统,记录远近探测器探测到的值,比较两种设计方案下远近探测器采集到的信号大小的比值,实验数据如图 4所示。

Figure 4. Ratio of light intensity received by near and far detectors with and without 1kΩ resistance a—cross 1kΩ resistance b—non span resistance
图 4中的数据表明,基于两种不同的电路设计方案,不同位置的光电探测器采集到数值的比值基本相同。实验表明,跨1kΩ电阻电路和利用光电探测器的内阻电路虽然输入电压不同,但放大倍数相同,输出的电压值均有良好的线性度,对实验数据没有影响。
-
上位机接收选用NI公司LabVIEW编写软件接收单片机发送的数据,进行数字滤波、运算并把结果以波形图的形式显示在界面中,程序如图 5所示。将光电探测器接收到的4路光强独立显示,以便监测实验中的数据变化。计算得到的RrSO2也以单独界面显示,做到连续、实时监测受试者血氧参量变化情况。
-
对受试者进行了前臂rSO2测量验证本系统的稳定性。由于该领域还没有公认的组织氧饱和度黄金标准[17],确定受试者身体健康状态良好的前提下进行测试,比对不同受试者之间的数据确定健康标准;对3名受试者进行长期测量,判断不同个体之间差异。皮瓣内血管栓塞的发生情况可以通过前臂静脉阻断模拟。为了能够无创监测人体皮肤内的血氧输运情况,利用袖带血压计在上臂施加一定的压力实现静脉阻断,从而测量前臂皮肤内的血氧变化情况。
为保证受试者测试时身体状态平稳,让受试对象在测试前放松10min,呼吸均匀、情绪稳定后进行测试。考虑到探测器与人体皮肤接触和光源自身发热导致受测组织局部温度上升,使得微血管内血液流速和血细胞的聚集状态发生改变影响实验的准确性[18]。因此,受试者的测量时间大于3min,等数值稳定后记录实验数据。
在一段时间内对受试者们集中测试,测试部位选择受试者的左前臂内侧、左前臂外侧、右前臂内侧、右前臂外侧。部分结果如表 1和图 6所示。
Table 1. Measurement value of tissue oxygen saturation
ID female(F)/
male(M)left forearm medial lateral left forearm right forearm medial lateral right forearm 1 F 0.536 0.528 0.524 0.527 2 M 0.500 0.505 0.512 0.526 3 M 0.530 0.504 0.545 0.490 4 F 0.517 0.510 0.525 0.492 5 F 0.531 0.505 0.524 0.502 6 M 0.530 0.520 0.527 0.510 7 F 0.538 0.523 0.542 0.520 8 M 0.528 0.519 0.539 0.521 9 M 0.524 0.472 0.532 0.474 10 M 0.508 0.481 0.501 0.502 11 F 0.531 0.515 0.538 0.531 12 F 0.508 0.484 0.526 0.496 13 F 0.520 0.497 0.523 0.517 14 F 0.536 0.533 0.534 0.520 15 M 0.530 0.512 0.509 0.506 16 M 0.543 0.480 0.528 0.515 17 F 0.521 0.525 0.526 0.515 18 F 0.526 0.504 0.516 0.492 19 M 0.510 0.509 0.510 0.524 20 M 0.525 0.493 0.538 0.497 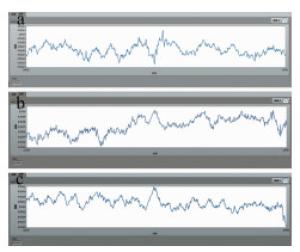
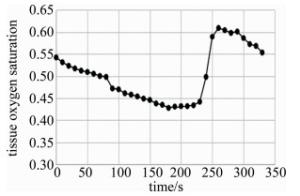
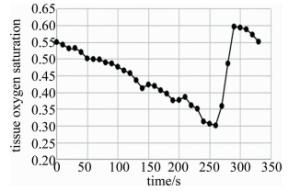
Figure 6. Long-term monitoring of three subjects a—long-term monitoring map of the first subject b—long-term monitoring map of the second subject c—long-term monitoring map of the third subject
数据表明不同个体、不同部位之间组织氧饱和度存在微小差异,幅度在0.05左右, 实验验证了本系统具有较高的稳定性, 这是因为不同受测者之间血管分布、脂肪厚度不同造成结果差异,符合客观事实。
对受试者进行静脉阻断实验来模拟皮瓣血管栓塞[19],阻断压力相同,阻断时间3min,测量结果如图 7、图 8所示。静脉阻断时,由于静脉回流受阻,CHBO2, CHB和血容积都迅速增加,此时手臂呈紫红色。一段时间后血液充盈减缓直至消失,此时rSO2才会随着皮肤耗氧开始缓慢减少。解除阻断后,由于血液循环恢复,rSO2快速回升并有过冲现象[20],之后逐步稳定到测量前的值。
-
由于临床术后护理大多依据主观经验判断皮瓣恢复情况,缺乏客观依据,基于近红外光谱技术,推导出双光源双探测器的测量算法并搭建了监测系统,进行了系统稳定性实验和静脉阻断模拟皮瓣栓塞实验,结果表明, 该系统稳定性良好,在静脉阻断下近红外光谱检测结果发生了显著改变,具有较高灵敏度,能够连续监测组织氧饱和度变化趋势,反映创面血液运输状况,为病人术后皮瓣血管的愈合程度提供客观依据。对辅助医生判断病人术后皮瓣存活状况有重要参考意义。
基于近红外光谱在皮瓣移植术后的监测系统
Monitoring systems for skin flap transplantation based on near infrared spectroscopy
-
摘要: 皮下组织血氧参量是反映皮瓣移植术后皮瓣存活状况的重要依据。为了无创、连续、实时地监测皮肤组织氧饱和度,采用近红外光谱法进行了理论分析和实验验证,提出了一种电流-电压(I-V)转换思路并搭建了测量系统。利用该系统进行了稳定性验证实验、前臂静脉阻断模拟皮瓣血管栓塞实验。结果表明,不同人体、不同部位的组织氧饱和度存在微小差异,差异幅度均在0.05左右;静脉阻断下系统组织氧参量发生显著改变,最大降幅0.25。该系统具有较高灵敏度,能够连续测量组织氧饱和度参量、反映组织氧变化趋势,可以为临床检测术后皮瓣血氧运输状态提供重要参考。Abstract: Oxygen parameters of subcutaneous tissue are the important basis to reflect the survival of skin flaps after skin flap transplantation. In order to monitor the oxygen saturation of skin tissue noninvasively, continuously and in real time, theoretical analysis and experimental verification were carried out by near infrared spectroscopy. A current-voltage (I-V) conversion method was proposed and a measurement system was built. The system was used to validate the stability and simulate the vascular embolization of forearm vein occlusion flaps.The results show that, there is a slight difference in tissue oxygen saturation in different parts of different human bodies, it is about 0.05. Under venous occlusion, oxygen parameters of the system tissue change significantly. The maximum decrease is 0.25. The system has high sensitivity. It can continuously measure tissue oxygen saturation parameters and reflect the trend of tissue oxygen change. It can provide important reference for clinical detection of oxygen transport status of skin flaps after operation.
-
Table 1. Measurement value of tissue oxygen saturation
ID female(F)/
male(M)left forearm medial lateral left forearm right forearm medial lateral right forearm 1 F 0.536 0.528 0.524 0.527 2 M 0.500 0.505 0.512 0.526 3 M 0.530 0.504 0.545 0.490 4 F 0.517 0.510 0.525 0.492 5 F 0.531 0.505 0.524 0.502 6 M 0.530 0.520 0.527 0.510 7 F 0.538 0.523 0.542 0.520 8 M 0.528 0.519 0.539 0.521 9 M 0.524 0.472 0.532 0.474 10 M 0.508 0.481 0.501 0.502 11 F 0.531 0.515 0.538 0.531 12 F 0.508 0.484 0.526 0.496 13 F 0.520 0.497 0.523 0.517 14 F 0.536 0.533 0.534 0.520 15 M 0.530 0.512 0.509 0.506 16 M 0.543 0.480 0.528 0.515 17 F 0.521 0.525 0.526 0.515 18 F 0.526 0.504 0.516 0.492 19 M 0.510 0.509 0.510 0.524 20 M 0.525 0.493 0.538 0.497 -
[1] TIAN F H, DING H Sh, WANG G Zh, et al. The use of near infra-red spectroscopy for monitoring perfusion and oxygen of skin[J]. Spectroscopy and Spectral Analysis, 2002, 22(2): 209-212(in Chinese). [2] LYU Ch F, LIU Sh D, ZHONG H, et al. Effect of hyperbaric oxygen therapy on the survival of skin flap after flap transplantation[J]. Chinese Journal of Aesthetic Medicine, 2019, 28(1): 31-33(in Chinese). [3] ZHANG Y Y. Nursing analysis of skin flap transplantation for hand wound repair[J]. World Latest Medicine Information, 2018, 18(94): 271-277(in Chinese). [4] DING H Sh, WANG G Zh, LI W, et al. Non-invasive quantitative assessment of oxidative metabolism in quadriceps muscles by near infrared spectroscopy[J]. British Journal of Sports Medicine, 2001, 35(6):441-444. doi: 10.1136/bjsm.35.6.441 [5] TENG Y Ch, DING H Sh, TIAN F H, et al. Measuring human tissue oxygenation with near infrared spectroscopy[J]. Journal of Tsinghua University(Science and Technology Edition), 2004, 44(6): 847-851(in Chinese). [6] SU Y, MENG Zh, YU H M, et al. Study on blood glucose in nonin-vasive measurement using optical coherence tomography[J]. Lsaer Technology, 2015, 39(1): 19-22(in Chinese). [7] LIU Y D, XU H, SUN X D, et al. Non-destructive measurement of tomato maturity bynear-infrared diffuse transmission spectroscopy[J]. Lsaer Technology, 2019, 43(1): 25-29(in Chinese). [8] DING H Sh, TENG Y Ch. Non-invasively monitoring human tissue oxygenation using near Infrared spectroscopy: Independent innovations[J]. Laser & Optoelectronics Progress, 2007, 44(9): 15-30(in Chinese). [9] MATCHER S J, ELWELL C E, COOPER C E, et al. Performance comparison of several published tissue near-infrared spectroscopy algorithms[J]. Anal Biochem, 1995, 227(1): 54-58. doi: 10.1006/abio.1995.1252 [10] YANG L, SUN J H. Photography measurement of optical parameters of biological tissues[J].Lsaer Technology, 2010, 34(6): 743-746(in Chinese). [11] ZHANG L Sh, ZHANG X H, WANG Zh Zh. Determination of optical coefficients of biological tissue by means of diffuse image[J].Lsaer Technology, 2011, 35(5): 675-677(in Chinese). [12] YANG L, GAO M T. Experimental study about measurement of optical parameters of biological tissue based on least square support vector machine[J]. Lsaer Technology, 2015, 39(3): 300-303(in Chinese). [13] FARRELL T J, PATTERSON M S. A diffusion theory model of spatially resolved steady-state diffuse reflectance for the noninvasive determination of tissue optical properties in vivo[J]. Medical Physics, 1992, 19(4): 879-888. doi: 10.1118/1.596777 [14] SUZUKI S, TAKASAKI S, OZAKI T. A tissue oxygenation monitor using NIR spatially resolved spectroscopy[J]. Proceedings of the SPIE, 1999, 3597: 582-592. doi: 10.1117/12.356862 [15] MATCHER S J, KIRKPATRICK P, NAHID K, et al. Absolute quantification methods in tissue near infrared spectroscopy[J]. Proceedings of the SPIE, 1995, 2389: 486-495. doi: 10.1117/12.209997 [16] TENG Y Ch, YE D T, LI Y, et al. Optimally designing the probe of the near infrared oximeter to detect human tissue oxygen saturation non-invasively[J]. Spectroscopy and Spectral Analysis, 2008, 28(4): 953-956(in Chinese). [17] DING H Sh, TENG Y Ch, LI Y. The noninvasive tissue oximeter using near infrared spectroscopy (NIRS) is first developed and clinically used in China[J]. Technology Report, 2005, 11(3): 30a-30d(in Chinese). [18] WU T H, XU K X, LIU Q Zh. A study of non-invasive measurement of human hemoglobin concentration by near infrared spectroscopy[J]. Acta Laster Biology Sinica, 2006, 15(2): 204-208(in Chinese). [19] YANG J R, QIN T, LI H W. An experimental study on processing microvascular circulation crisis of free skin flap[J]. Stomatology, 2002, 22(4): 172-173(in Chinese). [20] TIAN F H, DING H Sh, CAI Zh G, et al. Accessment of flap blood oxygen supply of rhesus using steady near infrared spectroscopy[J]. Chinese Science Bulletin, 2002, 47(16):1250-1255(in Chinese). doi: 10.1360/csb2002-47-16-1250 -

 网站地图
网站地图

 下载:
下载: